I first heard the term ‘angiosome’ on Tuesday the 27th of September 2022: Professor Standring was delivering her lecture ‘An Introduction to Anatomy’ in the Gordon Museum. She was discussing the history and future of anatomy and its influence on current medical practice: angiosomes were mentioned in the context of plastic and vascular surgery.
As a third-year medical student with a particular interest in anatomy and surgical ambitions, I was intrigued yet equally embarrassed I had never heard this term before. I decided to do some research. Here’s what I learnt.
What is an angiosome?
An angiosome is an anatomic unit of tissue, including skin, subcutaneous tissue, fascia, muscle and bone, fed by a source artery and drained by specific veins (1). It emphasises the three-dimensional nature of vasculature and describes the following characteristics:
Primary supply to skin from direct cutaneous arteries
Reinforced by small indirect vessels that tend to be terminal branches of arteries that primarily supply deeper tissues
Anastomotic arteries between adjacent angiosomes provide alternate supply if source artery is compromised
This concept was proposed by a plastic surgeon named G I Taylor in 1987. In a single word, he consolidated and integrated anatomical research and clinical knowledge that spanned decades.
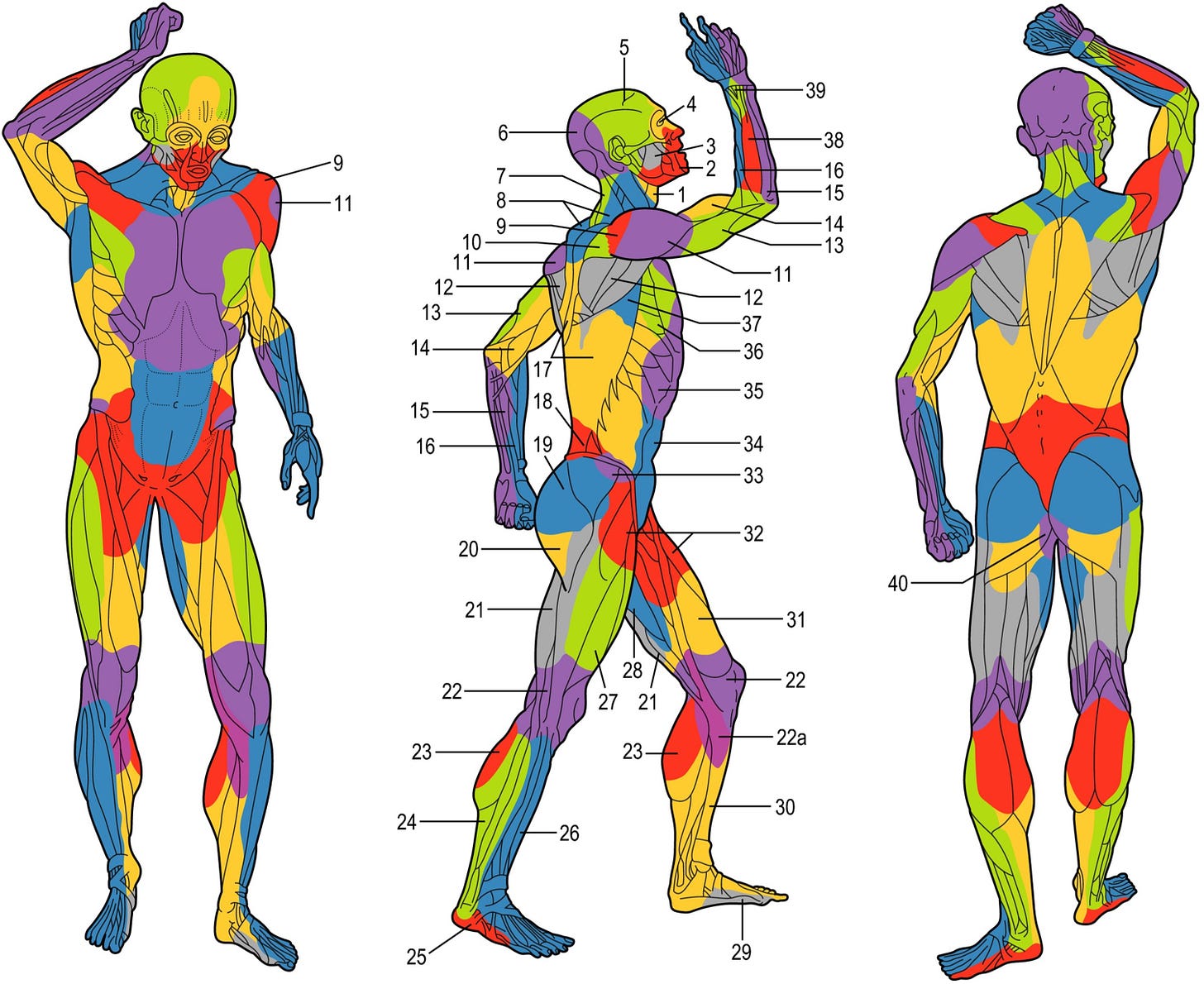
Figure 1 Angiosomes of the body (2)
The evolution of angiosomes
We will start in 1889 with the publication of Die Haut Arterien des Menschlichen Körpers – the Skin Arteries of the Body by Carl Manchot (3). Manchot mapped cutaneous perforating vessels to their underlying source vessels and plotted the cutaneous vascular territories of the body (Figure 2). Remarkably, Manchot achieved this feat in 6 months at 23 years-old without radiography: Wilhelm Röntgen was yet to discover his X-ray tube.
Michel Salmon followed in the 1930s and, this time with the help of X-rays, mapped not only the cutaneous circulation but also the blood supply to all the muscles in the body (4,5).
Figure 2 Carl Manchot’s vascular territories (3)
Progression on the clinical side offered further support for the angiosome concept. The artistry behind flaps pioneered by Sir Harold Gillies evolved to have rigid length to breadth ratios. Plastic surgeons started associating flaps with specific vasculature, for example a forehead flap and the superficial temporal vessels. Further work differentiated cutaneous and musculocutaneous vessels and identified that blood vessels follow fascial planes (6). Taylor began his work in 1970s, and following the dissection, injection and radiography of various regions of over 2000 cadavers he landed on the angiosome concept (1).
Figure 3 Montage of cutaneous circulation (1)
So, where does the angiosome concept sit presently?
The concept has garnered interest in the vascular surgery speciality in relation to revascularisation strategies: critical limb ischaemia represents the most severe form of Peripheral Arterial Disease (PAD) characterised by rest pain, ulcers or gangrene (7). Revascularisation via surgical bypass, or endovascular intervention, has traditionally utilised the ‘best vessel’ approach. The target outflow artery is chosen based on certain criteria including technical suitability, length of bypass required and disease characteristics, regardless of location (8,9). With reports of unhealed ischaemic wounds in up to 18% of cases leading to secondary amputation (8,10,11) a revascularisation strategy based on the angiosome concept has been suggested.
Applying the angiosome concept to revascularisation procedures would direct blood flow to areas of ischaemic ulceration via source artery’s rather than via the ‘best vessel’. Several techniques have been suggested enabling assessment of angiosome perfusion and adoption into revascularisation procedures.
Pre-operatively, handheld doppler devices can identify the source artery supplying relevant angiosomes and, with occlusion above and below the selected artery, reveal direction of flow (12).
Combining dye material and angiography yields more detailed angiosome vasculature anatomy mitigating the main limitation of using doppler: it does not provide direct visualisation of affected vasculature, which is often necessary for operational planning (13).
Fluorescent angiography lunges further. Injection of Indocyanine Green, a dye that absorbs in the near infrared spectrum and emits in the visible spectrum, is detected using a laser and allows immediate visualisation of tissue perfusion thereby offering real-time angiosome visualisation potential. Preliminary assessments are promising (14).
The angiosome concept underpins several appealing strategies in the context of revascularisation. This is particularly relevant with PAD rates on the rise. Currently in the Western world 1 in 5 adults over the age of 75 has PAD, however ageing populations and the increasing burden of chronic disease, including hypertension and diabetes, will further increase rates (15).
More research is warranted to determine significant differences in limb salvage and wound healing when comparing angiosome-based direct revascularisation and nonangiosome-based indirect revascularisation. Heterogeneity within the population makes this a particularly challenging task with variation in wound healing rates and collateral blood supply. Further limitations include technical feasibility and reproducibility between centres.
In conclusion, the angiosome concept offers an alternative approach to vascular, and plastic, pathologies. Adoption with new technologies potentiate its application further. However, further research is warranted and until then the ‘battle between blood supply and beauty’ will continue as noted by Gillies (16).
Taylor GI, Palmer JH. The vascular territories (angiosomes) of the body: experimental study and clinical applications. Br J Plast Surg. 1987 Mar;40(2):113–41.
Morris S, Taylor G. Vascular Territories. In: Plastic Surgery. PC Neligan. 2012. p. 479–511.
Manchot C. Die Hautarterien des menschlichen Körpers [Internet]. Vogel; 1889. Available from: https://books.google.co.uk/books?id=rqFMcgAACAAJ
Salmon M. Artères de la peau .. Paris: Masson et cie; 1936. 246 p.
Salmon M, Dor J. Les artères des muscles des membres et du tronc. Paris: Masson; 1933. 238 p.
Daniel RK, Williams HB. The free transfer of skin flaps by microvascular anastomoses. An experimental study and a reappraisal. Plast Reconstr Surg. 1973 Jul;52(1):16–31.
Novo S, Coppola G, Milio G. Critical Limb Ischemia: Definition and Natural History. Current Drug Targets - Cardiovascular & Hematological Disorders. 4(3):219–25.
Lepäntalo M, Biancari F, Tukiainen E. Never amputate without consultation of a vascular surgeon. Diabetes Metab Res Rev. 2000 Oct;16 Suppl 1:S27-32.
Sumpio BE, Lee T, Blume PA. Vascular evaluation and arterial reconstruction of the diabetic foot. Clinics in Podiatric Medicine and Surgery. 2003 Oct 1;20(4):689–708.
Berceli SA, Chan AK, Pomposelli FB, Gibbons GW, Campbell DR, Akbari CM, et al. Efficacy of dorsal pedal artery bypass in limb salvage for ischemic heel ulcers. J Vasc Surg. 1999 Sep;30(3):499–508.
Dorros G, Jaff MR, Dorros AM, Mathiak LM, He T. Tibioperoneal (Outflow Lesion) Angioplasty Can Be Used as Primary Treatment in 235 Patients With Critical Limb Ischemia. Circulation. 2001 Oct 23;104(17):2057–62.
Attinger CE, Evans KK, Bulan E, Blume P, Cooper P. Angiosomes of the Foot and Ankle and Clinical Implications for Limb Salvage: Reconstruction, Incisions, and Revascularization. Plastic and Reconstructive Surgery. 2006 Jun;117(7S):261S.
Yin ZX, Peng TH, Ding HM, Bai B, Tang ML. Three-dimensional visualization of the cutaneous angiosome using angiography. Clinical Anatomy. 2013;26(2):282–7.
Braun JD, Trinidad-Hernandez M, Perry D, Armstrong DG, Mills JL. Early quantitative evaluation of indocyanine green angiography in patients with critical limb ischemia. Journal of Vascular Surgery. 2013 May 1;57(5):1213–8.
Fowkes FGR, Rudan D, Rudan I, Aboyans V, Denenberg JO, McDermott MM, et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. The Lancet. 2013 Oct 19;382(9901):1329–40.
Gillies HD, Millard DR. The Principles and Art of Plastic Surgery. Little, Brown; 1957. 360 p.